COVID-19 and Aerogen

The last published Gold Science Committee Report for Covid-19 patients receiving respiratory support.;
"...It is vital to maintain circuit integrity and prevent transmission of the virus. The use of a Vibronik mesh nebulizer in ventilated patients enables drug delivery without compromising circuit integrity for aerosol drug therapy...."
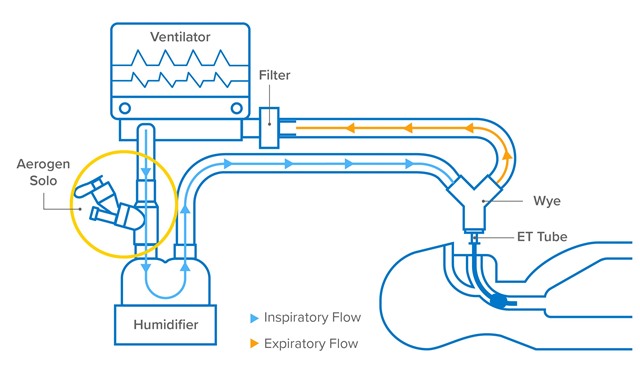
Aerogen, is the only closed circuit aerosol drug delivery system that reduces the contamination of the infectious Aerosol produced by the patient and provides effective aerosol therapy.
Discover that Aerogen‘s Vibronic Mesh technology can efficiently provide inhalation therapies for COVID-19 patients and is designed for the safety of both the patient and the healthcare worker. Numerous international articles have been published summarizing the use of this Mesh technology in the treatment of COVID-19 patients.
Aerogen is your solution to administer inhaled drugs to COVID patients.
Safe
Aerogen, is the only closed circuit aerosol drug delivery system that reduces contamination of the infectious aerosol generated by the patient.
Effective
Aerogen provides an effective inhalation drug. Times more medication is delivered to the lungs compared to a jet nebulizer.
Reliable
Aerogen, Dräger, GE, Hamilton Medical, Maquet, Medtronic and Philips many of the world‘s leading mechanical ventilation companies use and recommend the use of their ventilators integrated

International Academic Studies
International articles suggest that in order to deliver inhalation therapies to COVID-19 patients, the use of vibrating network technology to prevent transmission of the virus or the circuit should remain intact.
Academic Studies on Aerogen Use in COVID-19 Patients
WEB Seminars
During this time, some key opinion leaders took time to discuss Vibrating Mesh Technology and its use with COVID-19 patients. Findings on practical strategies for safe and effective aerosol drug therapy for patients with COVID-19; Dr Arzu Ari presented in his latest publication "Practical Strategies for a Safe and Effective Aerosolized Drug Treatment for Patients with COVID-19" on June 4, 2020.